If you’ve spent any time in a clinical oncology setting, you’ve likely seen the same frustrating pattern: a treatment that works wonders in a petri dish or a liquid tumor (like leukemia) suddenly hits a literal wall when faced with a solid mass.
At Immumem Therapeutics, we often talk about the “fortress” of the solid tumor. It’s not just a collection of runaway cells; it is a sophisticated, self-shielding ecosystem. For decades, the pharmaceutical industry has relied on conventional chemotherapeutic agents and large molecules (like monoclonal antibodies) to do the heavy lifting. But the reality is that these tools are often like trying to put out a fire inside a locked vault by spraying water on the outside of the building.
To build better treatments, we have to understand why the current ones are getting stuck at the gates.
The Architecture of Exclusion: Physical and Physiological Barriers
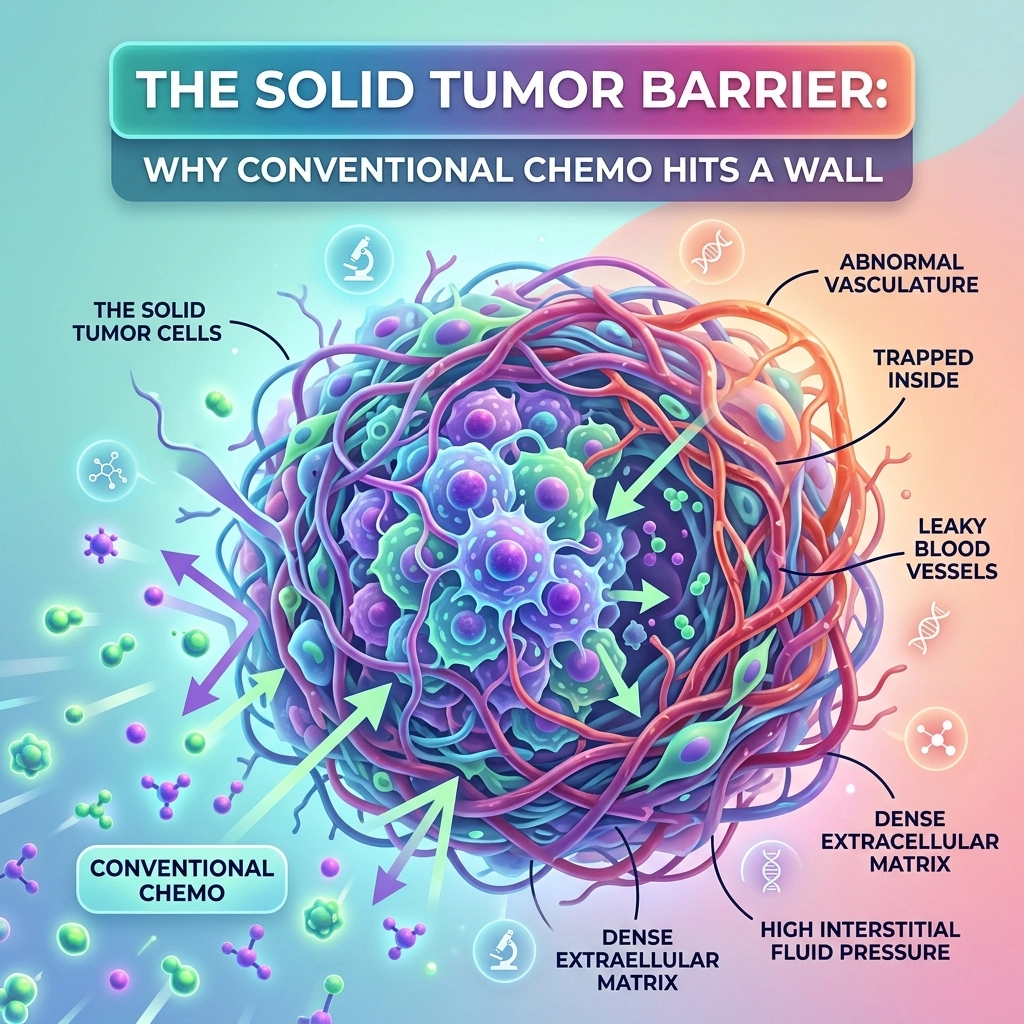
The primary reason conventional treatments fail in solid tumors isn’t always a lack of potency: it’s a lack of access. Solid tumors utilize a dual-layered defense system composed of structural (physical) and fluid-dynamic (physiological) barriers.
1. The Extracellular Matrix (ECM) “Tangle”
Think of the Extracellular Matrix as the “scaffolding” of the tumor. In healthy tissue, the ECM is a balanced network that provides structure. In a solid tumor, however, Cancer-Associated Fibroblasts (CAFs) go into overdrive. They remodel the ECM into a dense, collagen-rich thicket that is significantly stiffer than normal tissue.
According to research published in *Nature Communications*, this structural abnormality creates a mechanical barrier that restricts the diffusion of drugs. In aggressive cancers like pancreatic ductal adenocarcinoma, the stromal density is so extreme that it can actually collapse the local blood vessels, cutting off the “highway” that chemotherapy uses to arrive at the site. When the ECM is this dense, even small-molecule drugs struggle to navigate the maze, let alone larger biologic agents.
2. The Pressure Cooker: Interstitial Fluid Pressure (IFP)
In a healthy body, fluid moves from blood vessels into the surrounding tissue and is then drained away by the lymphatic system. In a solid tumor, the lymphatic “drains” are usually broken or non-existent. Coupled with “leaky” tumor blood vessels that dump fluid into the tumor core, this creates a phenomenon known as High Interstitial Fluid Pressure (IFP).
This pressure works as a literal outward force. As a drug tries to diffuse into the tumor, the internal pressure pushes it back out. It’s essentially a biological “no-entry” sign. This is a major reason why drug concentration at the center of a tumor is often negligible compared to the periphery, allowing the “core” of the cancer to survive and eventually trigger a relapse.
Why Large Molecules and Traditional Chemo Hit a Dead End
The pharmaceutical industry has leaned heavily on monoclonal antibodies (large molecules) because of their high specificity. However, their size is their greatest weakness in the context of solid tumors:
- Diffusion Limitations: Large molecules rely on passive diffusion to move through the tumor microenvironment (TME). Because of the high IFP and dense ECM mentioned above, the rate of diffusion for an antibody is exponentially slower than that of a small molecule. By the time an antibody might reach a cell in the tumor core, it may have already been cleared from the bloodstream or degraded.
- The “Binding Site Barrier”: This is a classic pharmacological paradox. When a large molecule drug is highly effective at binding to its target, it often binds to the very first cells it encounters at the edge of the tumor. This creates a “crust” of bound drugs that prevents further molecules from penetrating deeper.
- Conventional Chemotherapy’s Collateral Damage:While small-molecule chemo can sometimes penetrate better than antibodies, it lacks the “homing” capability. To get enough drug into the center of a dense tumor, you have to increase the systemic dose to levels that are often toxic to the patient’s healthy organs.
The result? Clinical data shows that the overall response rates to many immunotherapies and standard chemotherapies in solid tumors remain less than 30%. This gap in efficacy is precisely what next-generation immunotherapy aims to close.
Breaking the Wall: The Shift Toward iPSC-Derived Cell Therapy
If traditional drugs are “passive” travelers, then cell therapy represents an “active” infiltration force.
At Immumem Therapeutics, we are focusing on how we can engineer cells to not just find the tumor, but to actively move through these hostile environments. This is where **iPSC-derived cell therapy** (Induced Pluripotent Stem Cells) becomes a game-changer.
Unlike traditional CAR-T therapies that rely on harvesting a patient’s own (often exhausted) T-cells, iPSC technology allows us to create a standardized, “off-the-shelf” source of highly potent immune cells. We can “program” these cells with specific attributes to overcome the solid tumor barrier:
- Engineered Infiltration: We can equip these cells with synthetic fusion proteins or specific receptors that allow them to “digest” or navigate through the dense ECM.
- Resistance to Pressure: Unlike fluid-based drugs, living cells can move via chemotaxis: actively crawling toward chemical signals emitted by the tumor, regardless of the interstitial pressure.
- Persistence in the Microenvironment: The tumor microenvironment is often hypoxic (low oxygen) and acidic, which usually shuts down immune cells. Next-generation engineering allows us to create cells that thrive in these “dead zones.”
Beyond the Initial Kill: Immunological Memory and Relapse Prevention
Getting into the tumor is only half the battle. The most significant challenge in oncology isn’t just shrinking a mass; it’s ensuring it never comes back. This is the core of immunological memory oncology.
Conventional chemotherapy is a “one-and-done” event. Once the drug is cleared from the system, the protection is gone. If a single cluster of “persister cells” survives in the tumor core, they will eventually multiply, leading to a recurrence that is often more resistant than the original.
By leveraging cell therapies that promote long-term cancer relapse prevention, we are essentially “vaccinating” the patient against their own cancer. We want to engineer immune cells, specifically memory T-cells that remain in the body as “sentinels.”
- If a dormant cancer cell tries to wake up six months or six years after treatment, these memory cells are already there, primed and ready to neutralize the threat before it becomes a visible tumor.
- This shift from “treatment” to “surveillance” is the hallmark of the work we do at Immumem. You can see more about our approach on our overview page.
The Strategic Path Forward
For our partners in pharmaceutical companies and the investment community, the message is clear: the future of solid tumor treatment isn’t in making “stronger” chemicals, but in creating “smarter” cells.
The physical barriers of the tumor: the stiff ECM and the high fluid pressure: are insurmountable for 20th-century medicine. But they are merely engineering hurdles for 21st-century iPSC-derived cell therapy.
By focusing on immunological memory, we aren’t just looking for a higher Response Rate (RR); we are looking for a higher Cure Rate or at the least, turning a death sentence into a chronic manageable condition. We are moving toward a world where “remission” isn’t a temporary state, but a permanent one.
If you’re interested in the deep science behind how we’re re-engineering the immune system to scale these walls, we encourage you to dive into our Science Section or reach out to us directly via our contact page. We’re always looking for collaborators and visionaries who understand that to beat cancer, we have to stop hitting the wall and start moving through it.
Want to stay updated on our latest breakthroughs in immunological memory and solid tumor infiltration? Sign up for our newsletter here.